CagriSema Underperforms Zepbound: A Major Setback for Novo Nordisk in the Obesity Drug Race
The global race for dominance in the burgeoning obesity treatment market just witnessed a significant shift, as Novo Nordisk, a long-time leader, announced a major setback. Its much-anticipated next-generation obesity drug, CagriSema, has unfortunately underperformed Eli Lilly's formidable competitor, Zepbound (tirzepatide), in a crucial head-to-head trial. This unexpected blow to Novo Nordisk's ambitions has sent ripples through the pharmaceutical industry and left investors re-evaluating the competitive landscape.
Few analysts had predicted that the advanced combination drug from Novo Nordisk, positioned as a successor to its blockbuster Wegovy, would fall short against Eli Lilly's dual-action therapy. The revelation, based on recent cagrisema drug results, has not only cast a shadow over Novo Nordisk's future growth projections but has also solidified Eli Lilly's momentum in a market ripe with potential.
Understanding the Pivotal Trial and CagriSema Drug Results
The trial in question was designed with a clear objective: to demonstrate that CagriSema was at least as effective as tirzepatide in achieving significant weight reduction. Tirzepatide, known as Zepbound for weight loss in the U.S. and Mounjaro for diabetes and weight loss in other markets, has already established itself as a potent treatment. For Novo Nordisk, CagriSema was meant to reclaim or solidify its lead, especially with its existing drug Wegovy facing patent expiries post-2030.
However, the cagrisema drug results unveiled on Monday painted a different picture. Over an 84-week period, CagriSema achieved an average body weight reduction of 23%. While impressive in isolation, this figure was surpassed by Eli Lilly's tirzepatide, which demonstrated a 25.5% reduction. This seemingly minor difference of 2.5 percentage points in efficacy proved to be profoundly significant to both the scientific community and the investment world.
As Per Hansen of Nordnet aptly stated, "Weight loss of 23% versus 25.5% after 84 weeks may seem like a minor difference, but to investors it's very significant. In a winner-takes-all world, Eli Lilly has cemented its strong momentum." This sentiment highlights the fierce competition where even marginal differences can dictate market leadership and billions in revenue.
Market Reaction and Investor Impact
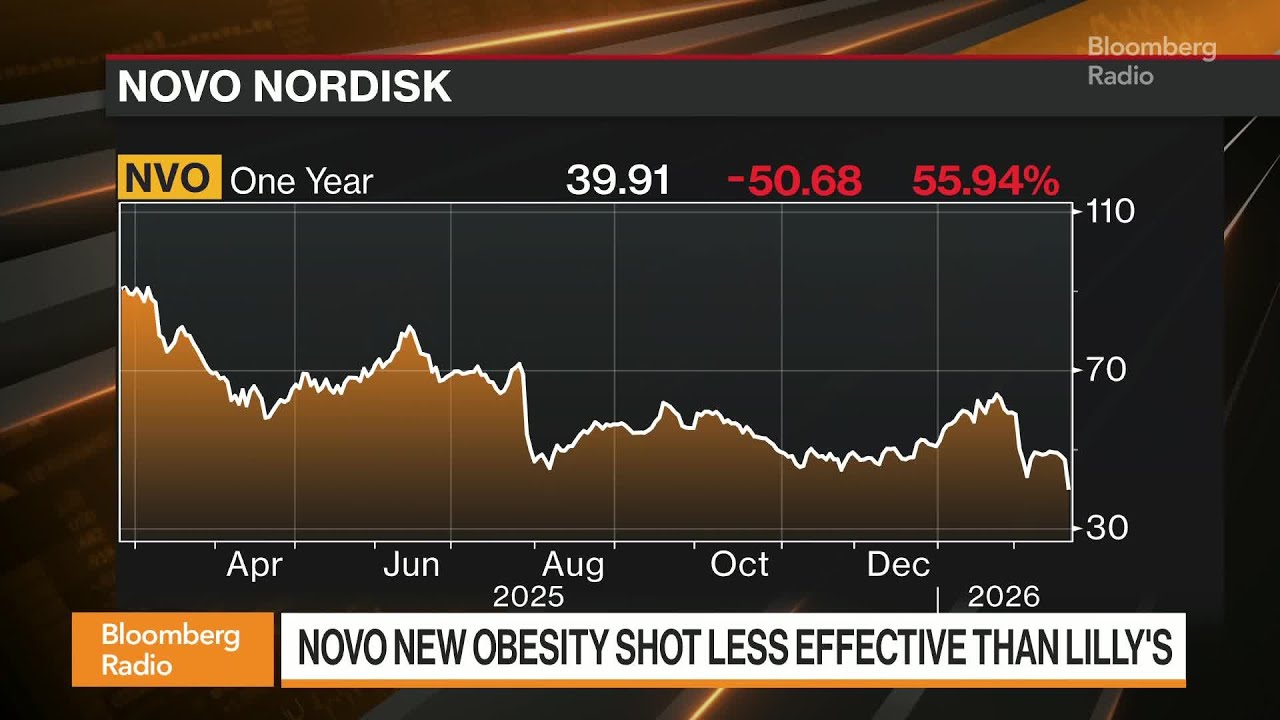
The immediate aftermath of the announcement was swift and severe for Novo Nordisk. The company's shares plummeted by 15%, reaching lows not seen since 2021. This sharp decline translated into a reduction of more than $400 billion from Novo Nordisk's market capitalization since its 2024 peak, effectively erasing the substantial gains accrued since the successful launch of Wegovy in 2021, which briefly positioned Novo Nordisk as Europe's most valuable listed firm.
This isn't the first time Novo Nordisk's shares have been impacted by cagrisema drug results. In December 2024, an earlier trial data release for CagriSema also led to a staggering $125 billion loss in market value in a single day. These events underscore the immense pressure and high stakes involved in the obesity drug market.
Conversely, Eli Lilly's shares experienced a rise in pre-market trading, reflecting the strengthened position of Zepbound and the positive outlook from investors on its continued market dominance. Analysts like Markus Manns at Union Investment, a shareholder in both companies, described the situation as a "worst-case scenario for Novo," acknowledging that it is now "clinically proven that Mounjaro is better than CagriSema." Henrik Hallengreen Laustsen of Jyske Bank echoed this, calling the news a "fairly big setback," especially considering that, according to consensus, CagriSema was expected to account for a substantial 60% of Novo Nordisk's future growth.
CagriSema's Scientific Profile and Previous Promise
CagriSema is a once-weekly injectable drug that combines two potent compounds: cagrilintide and semaglutide. Semaglutide is the active ingredient found in Novo Nordisk's existing weight loss drug, Wegovy, and mimics the gut hormone GLP-1 (glucagon-like peptide-1), which helps regulate appetite and blood sugar. Cagrilintide, on the other hand, mimics the pancreatic hormone amylin, further contributing to appetite control and glucose regulation. This dual-agonist approach was designed to offer a more comprehensive and potent solution for weight management.
Prior to this head-to-head trial, CagriSema had shown significant promise. Findings from two Phase 3 studies, REDEFINE 1 and REDEFINE 2, highlighted its potential for substantial weight loss in adults with and without type 2 diabetes who are overweight or obese. These 68-week studies, presented at the American Diabetes Association's 85th Scientific Sessions and published in the New England Journal of Medicine, indicated CagriSema's robust efficacy and safety profile. The REDEFINE 1 study, for instance, enrolled 3,400 adults without diabetes but with at least one weight-related health condition, demonstrating positive outcomes. These earlier cagrisema drug results had built considerable anticipation for its performance against direct competitors.
The recent trial, involving approximately 800 patients predominantly in the U.S., tested a fixed-dose combination of cagrilintide 2.4 milligrams and semaglutide 2.4 mg. While 23% weight loss is still a remarkable achievement for individuals struggling with obesity, the direct comparison with tirzepatide's 25.5% has unfortunately overshadowed this success in the competitive landscape.
The Evolving Obesity Drug Landscape: What's Next?
The outcome of this trial significantly reshapes the competitive dynamics of the obesity drug market. Eli Lilly's Zepbound has undeniably cemented its lead, establishing itself as the current benchmark for efficacy. This puts Novo Nordisk in an "uphill battle" with CagriSema, as analysts suggest. The company will now need to re-evaluate its strategy for the drug, potentially focusing on specific patient populations or emphasizing other benefits beyond sheer weight loss percentage, such as cardiovascular outcomes or tolerability profiles, that might differentiate it.
For patients, the continued innovation in this space is largely positive. While one drug may slightly outperform another, the availability of multiple highly effective treatments like Zepbound, Wegovy, and potentially CagriSema, offers more options for personalized care. The substantial weight loss achieved by both drugs—23% and 25.5%—represents life-changing results for many individuals struggling with obesity and its associated health conditions.
This intense competition also serves as a powerful catalyst for further research and development. Pharmaceutical companies will likely intensify their efforts to discover and develop even more effective and convenient obesity treatments, potentially exploring novel mechanisms of action or improved drug delivery systems. The race is far from over, but for now, Eli Lilly's Zepbound reigns supreme.
Conclusion
The latest cagrisema drug results represent a undeniable setback for Novo Nordisk in its quest to maintain market leadership in the booming obesity treatment sector. The underperformance against Eli Lilly's Zepbound, though by a seemingly small margin, has had profound financial implications and shifted investor confidence. While CagriSema remains a highly effective drug with significant potential for many patients, the competitive nature of the pharmaceutical market demands a "best-in-class" profile to truly dominate. As the industry moves forward, all eyes will be on how Novo Nordisk adapts its strategy and how this intense rivalry continues to drive innovation, ultimately benefiting the millions of individuals worldwide seeking effective solutions for weight management.